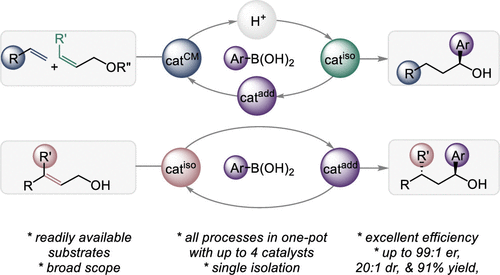
In this Letter, we report sequential multistep protocols with up to three transition-metal complexes and a Brønsted acid that execute redox-neutral transformations for a series of alkenes, (protected) unsaturated alcohols, and aryl boronic acids, with no or with minimal intermediary workup, to furnish varied secondary benzylic alcohols in high stereoselectivity, with up to 99:1 er, dr >20:1, and 91% yield. We showed that not only the protocols are operationally simpler and up to ∼3-fold less resource-intensive than the stepwise synthesis but also that the overall yield of the product is increased (77% versus 43%) thanks to preventing cumulative losses of the materials during subsequent isolations and purifications of the intermediates.
Congrats to all!
